Epidemiology
This page collects observations, interpretations, and consequences for action about the epidemiology of SARS-CoV2 in general. Please observe the structure of the page, when you add your content. Please use references where possible. Remember to find the relevant page. For example, if your observation is about transmission routes, please use that page, instead of posting your content here.
Contents
What is already known
- Coronavirus disease (COVID-19) is caused by SARS-COV2 and is a potentially fatal disease [1] that is designated a PHEIC by WHO [2].
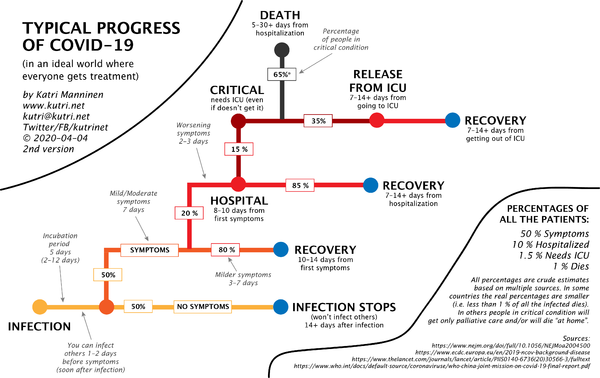
- Katri Manninen made the following infographic on the natural history of #COVID19: [3]
Symptoms
- The predominant symptoms in laboratory-confirmed cases is fever (88%) and dry cough (68%), followed by fatigue (38%), sputum production (33%), dyspnoea (19%), sore throat (14%), headache (14%) and myalgia or arthralgia (15%). Less common symptoms are diarrhoea (4%) and vomiting (5%).[4]
- About 80% of reported cases in China had mild to moderate disease (including non-pneumonia and pneumonia cases), 13.8% had severe disease and 6.1% were critical (respiratory failure, septic shock, and/or multiple organ dysfunction/failure). Current estimates suggest a median incubation period from five to six days for COVID-19, with a range from one to up to 14 days. [4]
- Italy reports consistently between 9% and 11% of the symptomatic cases to require intensive care treatment.[5]
Case fatality
- Reliable estimates for case fatality for COVID-19 are still lacking and biased. Based on a large dataset from cases in China, the overall case-fatality risk (CFR) among laboratory-confirmed cases was higher in the early stages of the outbreak (17.3% for cases with symptom onset from 1-10 January) and has reduced over time to 0.7% for patients with symptom onset after 1 February. In data on diagnosed COVID-19 cases in China, Italy and South Korea, overall CFR was 2.3%, 2.8% and 0.5%, respectively, and increased with age in all settings, with the highest CFR among people over 80 years of age (14.8%, 8.2% and 3.7%, respectively).[4]
Viral shedding
- Over the course of the infection, the virus has been identified in respiratory tract specimens 1-2 days before the onset of symptoms and it can persist for 7-12 days in moderate cases and up to 2 weeks in severe cases. [4]
- In faeces, viral RNA has been detected from day 5 after onset and up to 4 to 5 weeks in moderate cases. [4]
- SARS-CoV2 has been detected also in whole blood, serum, saliva and urine. [4]
- Prolonged viral RNA shedding has been reported from nasopharyngeal swabs, up to 37 days among adult patients and in faeces, for more than one month after infection in paediatric patients. [4]
Basic reproduction number (R0)
- The current estimates of the basic reproductive number R0 are between 2 and 3 in settings from China and during the early stage of an outbreak on a cruise ship. [4]
Asymptomatic Infections
- Based on Chinese data, the international WHO mission report indicates that up to 75% of initially asymptomatic cases will progress to clinical disease, making the true asymptomatic infection rather rare (estimated at 1-3%). [4]
- The virus has been detected in asymptomatic persons. On a rapidly evolving cruise ship outbreak, where most of the passengers and staff were tested irrespective of symptoms, 51% of the laboratory-confirmed cases were asymptomatic at the time of confirmation. In Italy, 44% of the laboratory-confirmed cases have been asymptomatic. In Japan, 0.06% of reported cases have been asymptomatic. These proportions based on nationally notified cases likely reflect laboratory testing algorithms rather than true estimates of asymptomatic infections. [4]
- Both viral RNA and infectious virus particles were detected in throat swabs from two German citizens evacuated from Hubei province on 1 February 2020, who remained well and afebrile seven days after admission to a hospital in Frankfurt. Both a mother and a child in a family cluster remained asymptomatic (including normal chest CT images during the observation period) with qRT-PCR positive nasopharyngeal swab samples. Similar viral load in asymptomatic versus symptomatic cases was reported in a study including 18 patients. Persistent positivity of viral RNA in throat and anal swabs were reported in an asymptomatic female patient after 17 days of clinical observation and treatment. [4]
- Potential transmission from an asymptomatic person has been reported in a familial cluster of five COVID-19 patients hospitalised with fever and respiratory symptoms that had contact before their onset of symptoms with an asymptomatic family member, a young 20-year-old woman, upon her return from Wuhan. She remained asymptomatic for the whole duration of laboratory and clinical monitoring (19 days).
- Transmission in a pre-symptomatic stage of infection: In addition to case reports, the pre-symptomatic transmission has been inferred through modelling, and the proportion of pre-symptomatic transmission was estimated to be around 48% and 62%. The pre-symptomatic transmission was deemed likely based on a shorter serial interval of COVID-19 (4.0 to 4.6 days) than the mean incubation period (five days) with the authors indicating that many secondary transmissions would have already occurred at the time when symptomatic cases are detected and isolated. Major uncertainties remain in assessing the influence of pre-symptomatic transmission on the overall transmission dynamics of the pandemic.
Vulnerable groups
- Data from Italy corroborate previously identified population groups at higher risk for having severe disease and death. These groups are elderly people above 70 years of age, and people with underlying conditions such as hypertension, diabetes, cardiovascular disease, chronic respiratory disease and cancer. Men in these groups appear to be at a higher risk than females. Chronic obstructive pulmonary disease (COPD), cardiovascular diseases, and hypertension have been identified as strong predictors for ICU admission.[6]
- Higher ACE2 (angiotensin converting enzyme II) gene expression may be linked to higher susceptibility to SARSCoV-2. It has been shown that ACE2 expression in lung tissues increases with age, tobacco use and with some types of antihypertensive treatment. These observations might explain the vulnerability of older people, tobacco users/smokers and those with hypertension; they also highlight the importance of identifying smokers as a potential vulnerable group for COVID-19. [6] [4]
- Currently available information indicates that children are as likely to be infected as adults, however they experience mild clinical manifestations. About 2.4% of the total reported cases in China (as of 20 February 2020) were individuals under 19 years of age. A very small proportion of those aged under 19 years have developed severe (2.5%) or critical disease (0.2%). [4]
Pregnant women and neonates
- A recent retrospective review of medical records by Chen et al. identified two cases of fetal distress among the nine monitored pregnancies, but did not identify any evidence of intrauterine infection. [7]
- Pregnant women appear to experience similar clinical manifestations as nonpregnant adult patients with COVID-19 pneumonia. There are only two reported cases of mothers with ICU admission and requiring mechanical ventilation or extracorporeal membrane oxygenation (ECMO). [6]
- No maternal deaths have been reported so far. COVID-19 appears to be less lethal for pregnant women than SARS (15% CFR in pregnancy) and MERS (27% CFR in pregnancy).[6]
- There is limited evidence of severe adverse outcomes, such as miscarriage, preterm birth, stillbirths and foetal distress. No pregnancy losses and only one stillbirth have been reported to date.[6]
- Intrauterine transmission appears to be unlikely. Elective Caesarean section deliveries have been commonly reported as a precautionary method to avoid perinatal transmission.[6]
- A confirmed COVID-19 neonatal case has been recently reported, however, the mode of transmission remains unclear. A neonate born to a confirmed maternal case had negative laboratory results for COVID-19 and died due to multi-organ failure.[6]
- The virus has not been found in breastmilk.[6] [4]
Children
- From early on, it was clear COVID19 attack rates in children were low. In Wuhan, no children tested positive between November 2019 and mid-January, per an early WHO report. [8]
- A study of 391 cases and 1,286 close contacts in Shenzhen, China, showed that kids under 9 had an attack rate of 7.4%, similar to adults (though adults 60-69 had a higher attack rate of 15.4%). [9]
- Another sophisticated study using multiple sources of data concluded that, compared to those aged 30–59 years, those aged <30 years and >59 years are 0.16 and 2.0 times more susceptible to symptomatic infection. [10]
- Transmission from pregnant women to their children in utero (known as ‘vertical transmission’) seems rare for COVID-19, too. [11] This is similar to SARS (from 2003).
- Not only is the symptomatic attack rate (mind you: not the Infection Rate) low in children (which we will need to confirm, eventually, with 'serological' tests in surveys), but the fatality rate, among those kids who do get infected, is very low.
- In the Diamond Princess cruise ship, there were 3,711 people and 634 cases of COVID19 (an attack rate of 17%). Half were asymptomatic. There were just 39 people <19 years old; only 5 got infected and only 2 had symptoms; and none died. [12]
- A large study of 2,143 pediatric patients in China found that, while 90% had mild or moderate illness, children <1 are more likely to have critical illness, if infected. [13]
- Overall, children are at a similar risk of infection as the adult population, though less likely to have severe symptoms, if infected. [9] [6]
- ECDC summarizes that "No evidence has been found to suggest that children are the primary drivers of SARS-CoV-2 virus transmission. However, research has shown that children can become infected, and spread the virus to adults while they are symptomatic." [14]
- Note that ECDC does NOT say that children do not contribute to community transmission, only that evidence is lacking that they are 'the primary driver'.
Reservoir and sources
- The three first reported cases were hospitalized on 27 December 2019 in Wuhan; one patient was a retailer on the seafood market in Wuhan, another was a frequent visitor of the same market. The third patients was not reported to be associated to the market. [15]
- COVID19 is likely of animal origin. [1] The virus has 85% similarity to a strain that was isolated from bats in East China. [15]
Transmission Routes
- Person-to-person transmission of COVID-19 infection is the predominant way in which this virus spreads among the human population. [1]. Transmission occurs through close contact (droplets, hands) and fomites. Therefore handwashing and social distancing are effective control measures, in the absence of a vaccine.
- Extensive measures to reduce person-to-person transmission of COVID-19 have been implemented to control the current outbreak. [1]
- Special attention and efforts to protect or reduce transmission should be applied in susceptible populations including children, health care providers, and elderly people.[1]
Detailed pages
For the following specific epidemiological topics, please use the subpages to document your observations:
For other epidemiological topics, please use this page.
Proportion asymptomatic
Observations
- There is clear evidence of asymptomatic infection with COVID-19. Estimates of the proportion of laboratory-confirmed cases who are asymptomatic may vary by age group, study setting and study methodology ranging from 1.2% (China)[16], 6.4% (Italy)[17], 12.9% (pediatric cases in China)[18], 13.0% (long-term care facility in Washington State) [19], and 17.9% (modelled estimate based on data from the Diamond Princess cruise ship)[20]. The extent to which screening is undertaken in group exposures and for contacts of cases, as well the duration of follow-up to ensure asymptomatic individuals do not subsequently develop symptoms will also influence the proportion of cases who are asymptomatic. [21]
- A cohort study around an outbreak of COVID19 in a bar in Vietnam in March 2020 suggests that the proportion of asymptomatic infections is larger than estimated so far. The study differentiated between asymptomatic (44% of all observed infections) and presymptomatic (38%). [22]
Analysis and interpretation
- The observations by Mizumoto and the International WHO Mission seemed to contradict on the issue of proportion asymptomatic infections. Though cruise ship populations are hardly representative of national populations, they can be considered reliable environments to study the natural history of infectious disease, when observing large groups of exposed susceptibles.
- The study by Valencia and others [22] is one of the few very well designed cohort studies in a field context where national quarantine guidelines required that close (high risk) contacts are quarantined in government facilities (instead of at home). This allows more reliable observations of the entire cohort of exposed people. Since everyone in the cohort is tested, there is not the usual selection bias (of only testing people with symptoms, or excluding certain ages). This study is also different from the Cruise Ship Studies, since the population in the study by Valencia has no homogenous mixing: After the exposure at the Bar on 14 March, all contacts were traced and quarantined. All infected contacts (apparent by positive test), were again subjected to contact-tracing, thus allowing a reliable observation of secondary attack rates for asymptomatic, pre-symptomatic and symptomatic cases.
- Having reliable estimates of the proportion asymptomatics is relevant for modelling the disease at the population level and making predictions about group immunity and transmission coefficients.
Consequences for action
- More field studies such as the one designed and performed by Valencia and others [22] are needed to further strengthen the body of evidence around the natural history of SARS-CoV2 infections. Understanding the extend of asymptomatic transmission is a relevant piece of evidence to be taken into account in response strategies (be it containment or mitigation)
Anosmia
Observations
- An increasing number of reports occur on the sudden loss of smell and taste among a proportion of the COVID19 patients. [23] [24] [25] [26] [27]
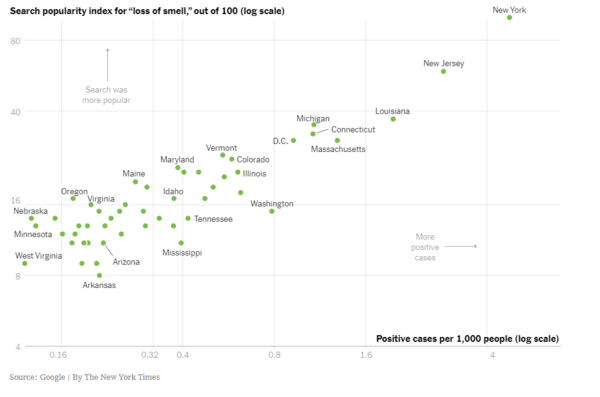
- Google analysis shows that 'I cannot smell' is a search term significantly associated with the locations where most COVID19 cases are reported. [28]
Analysis and Interpretation
- This observation requires a systematic clinical follow up of COVID19 patients, including pathognomonic studies
Consequences for Action
- If the relation COVID19 and Anosmia is confirmed, then clusters of sudden anosmia could be considered a clinical indicator of COVID19 in the population when testing is unavailable.
Unusual clinical expression
As in any novel pathogen, it may be difficult to fully understand the natural history of the disease, including the full spectrum of symptoms. Selection bias is always a risk, especially once we have agreed on a case definition that guides further case finding. In that sense, a case definition may become a 'self-fulfilling prophecy', since we filter out any unusual clinical manifestations. This section is used to report on observations.
Observations
- It seems that among children & adolescents, dermatological foot lesions are present, before the appearance of respiratory symptoms or fever. These are purple-coloured lesions, usually appear around the toes and heal without leaving marks on the skin.[29]
- Some note "chilblains-like lesion or as a vasculitis-like presentation on fingers and toes that can occur in children, teenagers and adults who have been diagnosed as positive for COVID-19 or are asymptomatic." [30]
- In Italy, health-care practitioners confirmed dozens of reports of acrocyanotic lesions in asymptomatic COVID-19-positive children and adolescents who were generally in good health prior to the skin rash onset. [31]
- Cardiac arrests and arrhythmias are likely the consequence of systemic illness and not solely the direct effects of COVID-19 infection. [32]
Analysis and interpretation
- The dermatological signs, if confirmed to be associated to SARS-CoV2, could be useful for identifying children and adolescents with minimal forms of infection, but potential sources of further infection.
Consequences for action
- We need to remain vigilant to unusual expressions of SARSCoV2 infections and not be caught in a tunnel vision as determined by strict COVID19 case definitions.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 Rothan, Hussin A., and Siddappa N. Byrareddy. "The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak." Journal of Autoimmunity (2020): 102433.
- ↑ Emergency committee regarding the outbreak of novel coronavirus(2019-ncov) https://www.who.int/news-room/detail/30-01-2020-statement-on-the-second-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-outbreak-of-novel-coronavirus-(2019-ncov)
- ↑ Katri Manninen on Twitter, April 2020: https://twitter.com/kutrinet/status/1246202064901939201?s=20
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 ECDC Rapid risk assessment: Novel coronavirus disease 2019 (COVID-19) pandemic: increased transmission in the EU/EEA and the UK – sixth update.
- ↑ COVID19 and Italy: what next? Remuzzi. Lancet, March 13 2020
- ↑ 6.0 6.1 6.2 6.3 6.4 6.5 6.6 6.7 6.8 ECDC Rapid Risk Assessment COVID19 7th Update. 25 March 2020.
- ↑ Zhang, Jiawei, et al. "Bioinformatic Analysis Reveals That the Reproductive System is Potentially at Risk from SARS-CoV-2." (2020).
- ↑ Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19) 16-24 February 2020.
- ↑ 9.0 9.1 Bi, Q., Wu, Y., Mei, S., Ye, C., Zou, X., Zhang, Z., ... & Gao, W. (2020). Epidemiology and Transmission of COVID-19 in Shenzhen China: Analysis of 391 cases and 1,286 of their close contacts. medRxiv.
- ↑ Wu, J. T., Leung, K., Bushman, M., Kishore, N., Niehus, R., de Salazar, P. M., ... & Leung, G. M. (2020). Estimating clinical severity of COVID-19 from the transmission dynamics in Wuhan, China. Nature Medicine, 1-5.
- ↑ Dong L, Tian J, He S, et al. Possible Vertical Transmission of SARS-CoV-2 From an Infected Mother to Her Newborn. JAMA. Published online March 26, 2020. doi:10.1001/jama.2020.4621
- ↑ Russell, T. W., Hellewell, J., Jarvis, C. I., van-Zandvoort, K., Abbott, S., Ratnayake, R., ... & CMMID nCov working group. (2020). Estimating the infection and case fatality ratio for COVID-19 using age-adjusted data from the outbreak on the Diamond Princess cruise ship. medRxiv.
- ↑ Dong, Y., Mo, X., Hu, Y., Qi, X., Jiang, F., Jiang, Z., & Tong, S. (2020). Epidemiological characteristics of 2143 pediatric patients with 2019 coronavirus disease in China. Pediatrics.
- ↑ Q&A on COVID-19 in children aged 0–18 years and the role of school settings in COVID-19 transmission. https://www.ecdc.europa.eu/en/covid-19/facts/questions-answers-school-transmission Webpage, accessed 18 September 2020.
- ↑ 15.0 15.1 Zhu, Na, et al. "A novel coronavirus from patients with pneumonia in China, 2019." New England Journal of Medicine (2020).
- ↑ Surveillances, V. (2020). The Epidemiological Characteristics of an Outbreak of 2019 Novel Coronavirus Diseases (COVID-19)—China, 2020. China CDC Weekly, 2(8), 113-122.
- ↑ Integrated surveillance of COVID19 in Italy. Website ISS
- ↑ Dong, Y., Mo, X., Hu, Y., Qi, X., Jiang, F., Jiang, Z., & Tong, S. (2020). Epidemiological characteristics of 2143 pediatric patients with 2019 coronavirus disease in China. Pediatrics.
- ↑ Kimball, A. (2020). Asymptomatic and Presymptomatic SARS-CoV-2 Infections in Residents of a Long-Term Care Skilled Nursing Facility—King County, Washington, March 2020. MMWR. Morbidity and Mortality Weekly Report, 69.
- ↑ Mizumoto, K., Kagaya, K., Zarebski, A., & Chowell, G. (2020). Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020. Eurosurveillance, 25(10).
- ↑ What We Know So Far About… Asymptomatic Infection and Asymptomatic Transmission. SYNOPSIS Version 4 03/30/2020 COVID-19 – Public Health Ontario
- ↑ 22.0 22.1 22.2 Valencia C, Quang L, Handcock M, Nguyen D, Doan Q, Nguyen TV, Le N, Truong T, Do H, Otsu S, Le T. Asymptomatic and Presymptomatic Transmission of 2019 Novel Coronavirus (COVID-19) Infection: An Estimation from a Cluster of Confirmed Cases in Ho Chi Minh City, Vietnam.
- ↑ If you’ve lost your sense of smell or taste, you could be a ‘hidden carrier’ of the coronavirus. Brianna Moné. Business Insider 22 Mar 2020
- ↑ Brann, D., T. Tsukahara, C. Weinreb, and D. W. Logan. 2020. “Non-Neural Expression of SARS-CoV-2 Entry Genes in the Olfactory Epithelium Suggests Mechanisms Underlying Anosmia in COVID-19 Patients.” bioRxiv. https://trafficlight.bitdefender.com/info?url=https://www.biorxiv.org/content/10.1101/2020.03.25.009084v1.abstract.
- ↑ Iacobucci, Gareth. 2020. “Sixty Seconds on... Anosmia.” BMJ 368 (March): m1202.
- ↑ Bagheri, Seyed Hamid Reza, Ali Mohammad Asghari, Mohammad Farhadi, Ahmad Reza Shamshiri, Ali Kabir, Seyed Kamran Kamrava, Maryam Jalessi, et al. 2020. “Coincidence of COVID-19 Epidemic and Olfactory Dysfunction Outbreak.” medRxiv. https://trafficlight.bitdefender.com/info?url=https://www.medrxiv.org/content/10.1101/2020.03.23.20041889v1.abstract.
- ↑ Hopkins, C., and N. Kumar. 2020. “Loss of Sense of Smell as Marker of COVID-19 Infection.” Retrieved from: https://www.entuk.org/sites/default/files/files/Loss%20of%20sense%20of%20smell%20as%20marker%20of%20COVID.pdf
- ↑ Google Searches Can Help Us Find Emerging Covid-19 Outbreaks. They can also reveal symptoms that at first went undetected. I may have found a new one. New York Times. 5 April 2020
- ↑ Anekdotal an observation shared by Grazia Caleo, MSF, on 16 April 2020
- ↑ COVID-19 Skin Manifestations And The Foot: What We Know So Far. April 22, 2020 By Tracey Vlahovic DPM. https://www.podiatrytoday.com/blogged/covid-19-skin-manifestations-and-foot-what-we-know-so-far
- ↑ Mazzotta F, Troccoli T. Acute acro-ischemia in the child at the time of COVID-19. International Federation of Podiatrists. Available at: https://www.fip-ifp.org/wp-content/uploads/2020/04/acroischemia-ENG.pdf Accessed April 16, 2020.
- ↑ Bhatla, A., Mayer, M. M., Adusumalli, S., Hyman, M. C., Oh, E., Tierney, A., ... & Domenico, C. M. (2020). COVID-19 and Cardiac Arrhythmias. Heart Rhythm.